North Carolina Couple Receives New Infusion Treatment for COVID-19 at McLeod
Treatment Reduces the Disease Progression of COVID-19
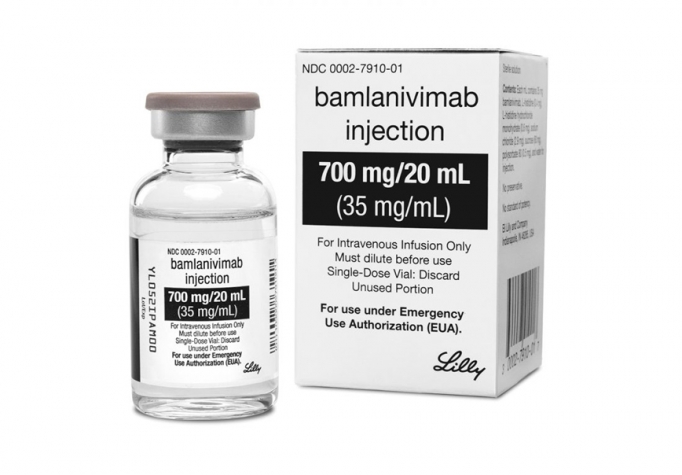
McLeod Health is pleased to announce it is offering the monoclonal antibody therapy Bamlanivimab as an outpatient infusion treatment for patients at high risk of developing COVID-19.
Myrtle and Richard Rudzinski of Carolina Shores, North Carolina, were two of the first few patients infused at McLeod Regional Medical Center the week of November 23. A week later, Richard reported that they were both doing well and feeling better each day.
“We are extremely grateful for everything McLeod Health did for us as we had no idea this awesome treatment was available. Our infusion nurse Christina was like an angel and did her best to make us feel comforted. She is also very good at her job. We truly want to thank everyone at McLeod for our speedy recovery at a critical time,” said Richard.
The one-time intravenous therapy which received Emergency Use Authorization from the FDA on November 9, 2020, is designed to prevent patients like the Rudzinskis from being hospitalized for the virus.
Bamlanivimab is a new investigational medicine used for the treatment of COVID-19 in non-hospitalized adults with mild to moderate symptoms and who are at high risk for developing severe COVID-19 symptoms or the need for hospitalization. High risk patients include those 65 or older, who have a BMI of 35 or more, or who are 55 or older with hypertension, heart disease, or respiratory disease.
McLeod Health is screening patients who are positive for COVID-19 for possible treatment each day and contacting these patients with information about the therapy. The medication is a 1 hour IV infusion, followed by a 1 hour monitoring period. An appointment lasts roughly 3 hours and the known side effects are rare.
McLeod Regional Medical Center in Florence began treating patients with Bamlanivimab on November 24, 2020. The treatment will also soon be available at McLeod Health Seacoast in Little River and McLeod Health Clarendon in Manning.
-
McLEOD REGIONAL MEDICAL CENTER FLORENCE
843-777-2000 -
McLEOD DARLINGTON
843-777-1100 -
McLEOD DILLON
843-774-4111 -
McLEOD LORIS
843-716-7000 -
McLEOD SEACOAST
843-390-8100 -
McLEOD CHERAW
843-537-7881 -
McLEOD CLARENDON
803-433-3000


-
McLEOD REGIONAL MEDICAL CENTER FLORENCE
843-777-2000 -
McLEOD DARLINGTON
843-777-1100 -
McLEOD DILLON
843-774-4111 -
McLEOD LORIS
843-716-7000 -
McLEOD SEACOAST
843-390-8100 -
McLEOD CHERAW
843-537-7881 -
McLEOD CLARENDON
803-433-3000
 Find a Doctor
Find a Doctor  Locations
Locations  Services
Services